PC 03
POLYANILINE NANOTUBES PREPARED IN SOLUTIONS OF VARIOUS ACIDS
E.N. Konyushenko, J. Stejskal, M. Trchová
Institute of Macromolecular Chemistry, Academy of Sciences of the Czech Republic, 162 06 Prague 6, Czech Republic (elenak@imc.cas.cz)
Polyaniline is the most attractive conducting polymer due to low cost of preparation and interesting redox and ion-exchange properties. The oxidation of aniline has been carried out at different acidity conditions. Aniline was polymerized in the aqueous solutions of strong (sulfuric) or weak (acetic, succinic) acids, and in the absence of any acid.1
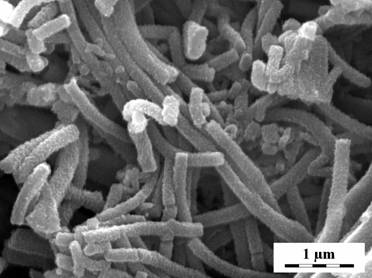
 Granular structure was obtained when aniline was oxidized in the presence of strong acid, like sulfuric acid. Polyaniline nanotubes were synthesized when weak organic acids were used, like succinic or acetic acids (Fig. 1). Polyaniline nanotubes were also obtained when aniline was oxidized in the absence of any acid.2 The molecular structure of nanotubes was studied by Raman and FTIR spectroscopies.3 The explanation of nanotubes formation is still a challenge. It is proposed that aniline oligomers containing phenazine constitutional units act as nucleation templates for the nanotubular growth of polyaniline. 2,3
Granular structure was obtained when aniline was oxidized in the presence of strong acid, like sulfuric acid. Polyaniline nanotubes were synthesized when weak organic acids were used, like succinic or acetic acids (Fig. 1). Polyaniline nanotubes were also obtained when aniline was oxidized in the absence of any acid.2 The molecular structure of nanotubes was studied by Raman and FTIR spectroscopies.3 The explanation of nanotubes formation is still a challenge. It is proposed that aniline oligomers containing phenazine constitutional units act as nucleation templates for the nanotubular growth of polyaniline. 2,3
References
1. E. N. Konyushenko, J. Stejskal, I. Šeděnková, M. Trchová, I. Sapurina, M. Cieslar, J. Prokeš, Polym. Int. 2006, 55, 31.
2. J. Stejskal, I. Sapurina, M. Trchová, E. N. Konyushenko, P. Holler, Polymer 2006, 47, 8253.
3. M. Trchová, I. Šeděnková, E. N. Konyushenko, J. Stejskal, P. Holler, G. Ćirić-Marjanović, J. Phys. Chem. B 2006, 110, 9461.